Element
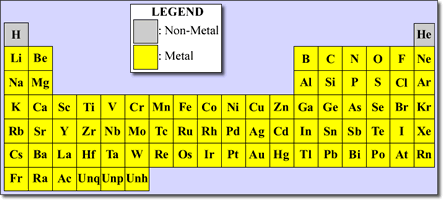
The Periodic Table lists elements in ascending atomic number.
An element is a material or substance made from atoms that have the same atomic number. For example oxygen is an element that is composed of oxygen atoms (atoms with eight protons, electrons and neutrons).
Elements may undergo chemical reactions with other elements or compounds to form new compounds. There are more than 110 known elements, with new ones still being discovered – this is because some elements are very unstable and cannot exist except under special conditions (the heaviest stable element is uranium). These unstable transuranic elements can be created by colliding heavy nuclei together.
The elements are most commonly arranged in ascending atomic number (Z) order in the Periodic Table.
Study Astronomy Online at Swinburne University
All material is © Swinburne University of Technology except where indicated.

