Nucleosynthesis in stars
Some basic facts of nuclear physics An atomic nucleus consists of Z protons and N neutrons, where Z is the atomic number defining the charge of the nucleus, the number of electrons in the neutral atom and hence the chemical element, and Z + N = A, the mass number of the nuclear species. Different values of A and N for a given element lead to different isotopes, while nuclei with the same A and different Z are referred to as isobars.
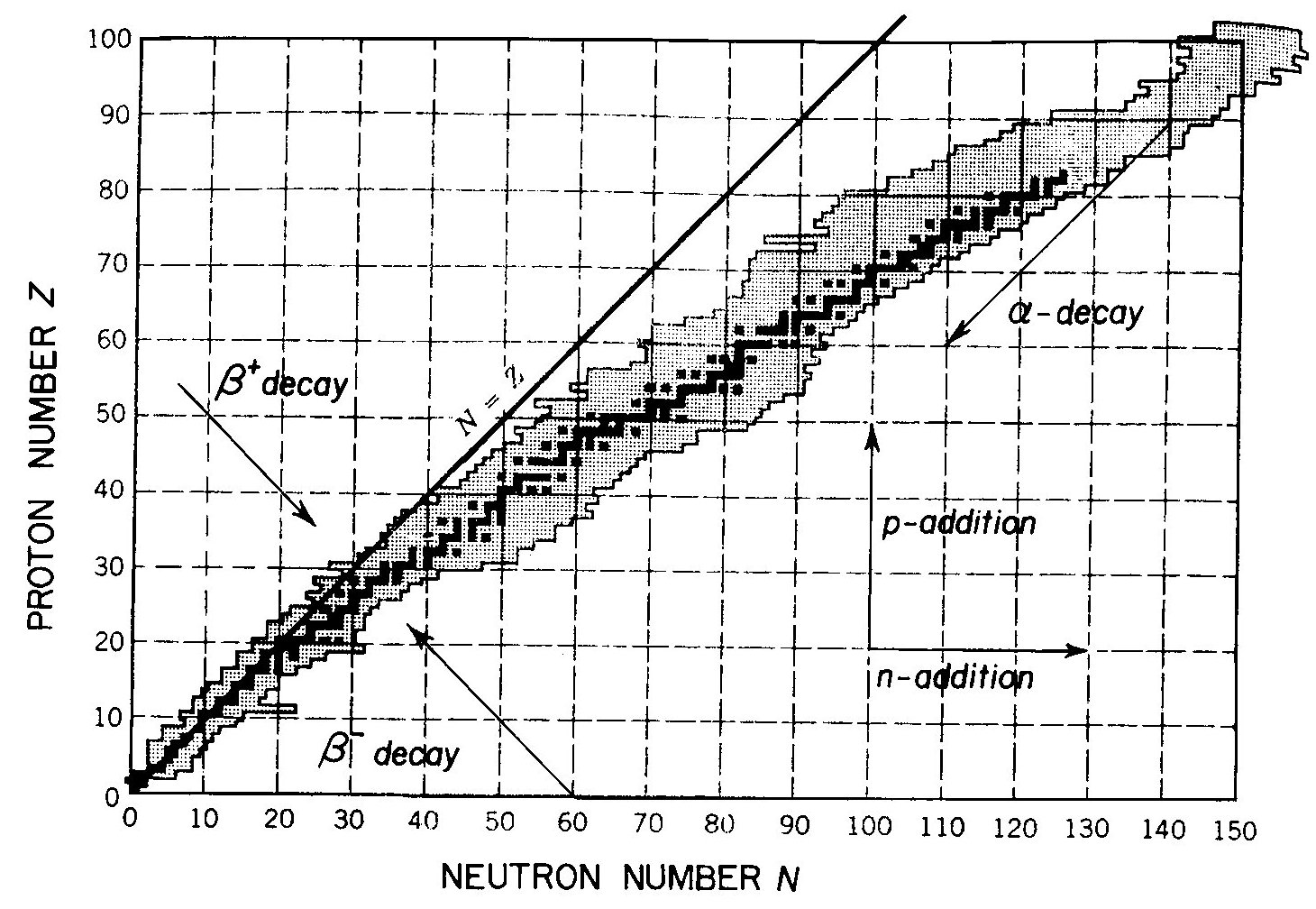
Stable nuclei occupy a ‘β-stability valley’ in the Z, N plane (see Figure below). Various processes, some of which are shown in the figure, transform one nucleus into another. Thus, under normal conditions, a nucleus outside the valley undergoes spontaneous decays, while in accelerators, stars and the early Universe nuclei are transformed into one another by various reactions.
Looking at the chemical abundances of a Sun main sequence star (a detailed description of stellar evolution can be found on Stellar Evolution)
| Abundances | ||
|---|---|---|
| Element | Young Stars | Sun |
| Hydrogen (X) | 0.6 | 0.7 |
| Helium (Y) | 0.36 | 0.2 |
| Metals (Z) | 0.04 | 0.02 |
We can notice that Hydrogen is the most element in the Stars (and in the Universe), and it's from this element that stars begin their life on the Main Sequence by the different PPchains (Proton-Proton Chains):
PPI chain
H1 + H1 &rarr D2 + e+ + ν
D2 + H1 → He3 + γ
He3 + He3 → He4 + 2H1
or with a lower probability
| PPII chain | PPIII chain |
| H1 + H1 &rarr D2 + e+ + ν | H1 + H1 &rarr D2 + e+ + ν |
| D2 + H1 → He3 + γ | D2 + H1 → He3 + γ |
| He3 + He4 → Be7 + γ | He3 + He4 → Be7 + γ |
| Be7 + e- → Li7 + ν | Be7 + H1 → B8 + γ |
| Li7 + H1 → He4 + He4 | B8 → Be8 + e+ + ν |
| Be8 → He4 + He4 |