Ion
An ion is formed when an atom loses or gains electrons. This causes an otherwise neutral atom to acquire a charge:
- a positively charged cation is created if electrons are lost.
- a negatively charged anion is created if electrons are gained.
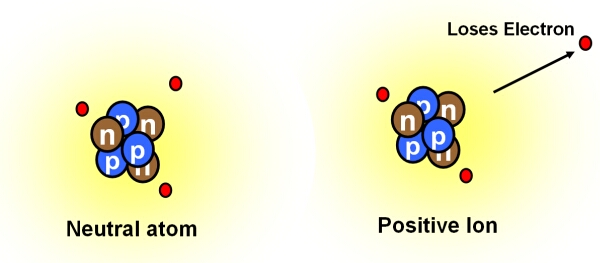
A neutral atom loses an electron to become a positively charge ion (cation).
The ease with which an atom or molecule can be ionised depends on the distribution of electrons.
Study Astronomy Online at Swinburne University
All material is © Swinburne University of Technology except where indicated.

